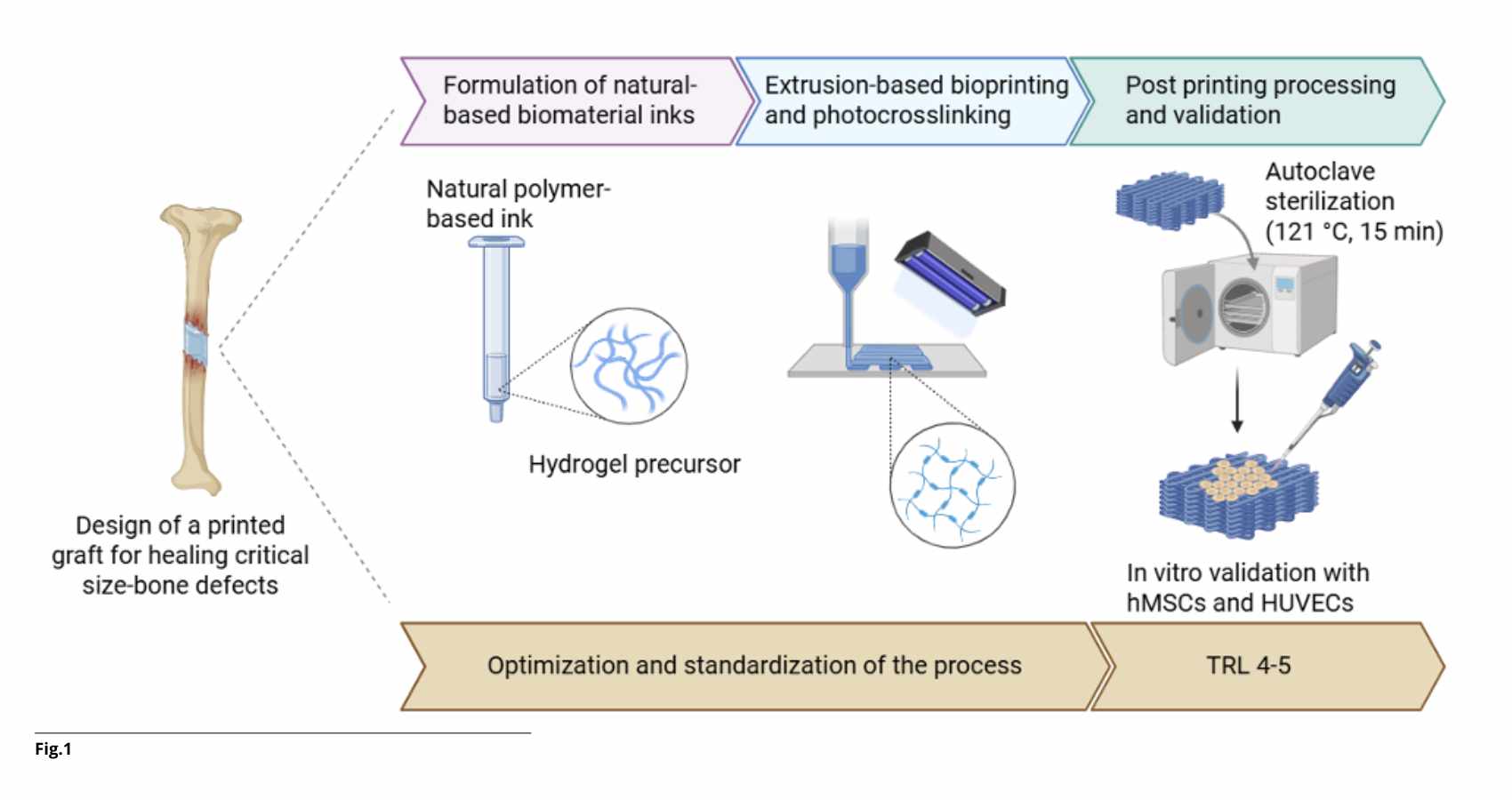
The human body’s ability to self-repair is remarkable, but not unlimited. When a portion of bone is extensively damaged (critical-size bone defects) – for example due to severe trauma, tumors, or degenerative diseases – natural regeneration is no longer sufficient. In these cases, reconstructing bone remains one of the most challenging tasks for orthopedic and maxillofacial surgeons.
Bone grafts, i.e., portions of bone taken from the patient or from a donor, have long represented the gold standard treatment, but they present significant limitations, including limited donor tissue availability and the risk of complications.
For this reason, in recent years research has focused on Bone Tissue Engineering, a field aimed at creating bone substitutes in the laboratory: porous three-dimensional structures (scaffolds) that act as temporary frameworks to guide new tissue formation and gradually degrade over time.
One of the most promising technologies to produce these structures is 3D bioprinting, which enables the fabrication of three-dimensional constructs with extremely precise spatial control over the deposition of biomaterials, bioactive molecules, and patient-derived cells. This makes it possible to replicate the hierarchical architecture and micro-porosity of native bone, overcoming the geometric limitations of conventional techniques.
The main challenge today is identifying suitable bioinks: materials that can be easily printed while simultaneously providing an ideal environment for cells.
This research originates from an international collaboration between the BIOtech Center of the Department of Industrial Engineering at the University of Trento (under the supervision of Prof. Antonella Motta and Prof. Devid Maniglio) and the Charles Perkins Centre at the University of Sydney, Australia (under the supervision of Prof. Anthony S. Weiss).
The project is part of the European MSCA-RISE SHIFT program (Shaping Innovative Products for Sustainable Tissue Engineering Strategies), which focuses on the use of natural materials to develop scalable and sustainable solutions in regenerative medicine.
The international experience confirmed how global collaboration is a cornerstone of scientific research: interaction with different academic environments not only accelerates experimental results but also represents a fundamental step in the professional development of researchers.
The starting material of this study was silk fibroin (SF), a natural polymer already known for its biocompatibility and relatively good mechanical properties. However, on its own, it is not easily printable: it does not form a stable filament nor maintain its shape after deposition.
To overcome these limitations, a multiscale design approach was adopted, ranging from nanometric chemical functionalization to micrometric rheological modulation, up to macroscopic structural validation.
Printability was optimized by blending silk fibroin with methacrylated hyaluronic acid (HAMA). Thanks to its high viscosity and hydrophilicity, HAMA imparts shear-thinning behavior to the system: the ink flows under pressure during extrusion and rapidly recovers its viscosity at rest, ensuring the necessary shape fidelity.
In parallel, a physical pre-crosslinking strategy using ultra-short self-assembling peptides was introduced, enhancing the viscoelastic properties of the material prior to printing. The synergistic integration of HAMA and peptides enabled optimal shape fidelity.
Once printability was optimized, the research focused on another key aspect: bioactivity specific to bone tissue. The incorporation of nano-hydroxyapatite (HAP) – the main mineral component of bone – transformed the hydrogel into a biomimetic composite, improving its mechanical stability and its ability to promote adhesion and differentiation of human mesenchymal stem cells (hMSCs).
During the research period in Sydney, the scaffolds (the printed structures) were further improved by coating them with tropoelastin, the precursor of elastin, which promoted the formation of new blood vessels and bone tissue growth without altering the material’s mechanical properties.
In line with the sustainability principles of the SHIFT project, a green chemistry strategy was also adopted by replacing synthetic hydroxyapatite with a biogenic variant obtained from mussel shells—food waste valorized through mechano-chemical synthesis. In vitro tests showed higher metabolic activity in samples containing the natural variant, demonstrating that sustainability and biological performance can go hand in hand.
Another objective of the research was to make the printing process more reliable and reproducible. Indeed, the lack of standardization is one of the main obstacles to the clinical translation of bioprinting. A method for standardizing the extrusion process was therefore developed, based on predictive mathematical models of printing parameters. This contribution aims to strengthen experimental reproducibility and to integrate current standards, such as ASTM F3659.
Overall, this work demonstrates that it is possible to produce 3D-printed, bioactive, and customizable bone structures using natural materials.
The rational integration of silk fibroin, hyaluronic acid, hydroxyapatite, and tropoelastin made it possible to simultaneously meet mechanical, rheological, and biological requirements, paving the way for solutions that can truly be translated from research to clinical practice.
The direction is clear: bringing these solutions from the laboratory to the operating room. This is the trajectory of a line of research showing how natural materials, engineering expertise, and international collaboration can converge toward new frontiers in regenerative medicine.
Fig.3: On the left, an example of 3D bioprinting of natural polymers; on the right, interaction of stem cells (nuclei in blue and cytoskeleton in green) with the scaffold optimized in this project.